

Document Type : Original Article
Authors
1 Urology Research Center,Tehran University of Medical Sciences, Tehran, Iran
2 Department of Gynecological Oncology, Mirza Koochak Khan Hospital, Tehran, Iran
3 Department of Biomadical Engineering, Amirkabir University of Technology, Tehran, Iran
4 Department of Medicine, Tehran University of Medical Sciences, Tehran, Iran
Abstract
Highlights
Keywords
Introduction
The prevalence of urinary stones ranges from 2 to 3 percent in different communities (1). Calcium stones comprise 80 to 85 percent of urinary stones (2). The exact cause of the formation of these stones is unknown. Researches show that about 90 percent of patients with urinary stones have some form of metabolic disorder. These disorders involved in developing urinary stones include hypercalciuria, hyperuricosuria, hyperoxaluria, and hypocitraturia (3).
Fass et al., (4) showed that patients with urinary tract stones had a significantly lower bone mineral content (BMC) than healthy subjects. The exact cause of this phenomenon is unclear. However, it is obvious that patients with urinary tract stones also have severe skeletal problems. Primary disorder in bone metabolism is suggested as one of the mechanisms that explain bone density reduction in patients with a low calcium diet with urinary tract deficiency.
Since no research has been done on the correlation of urinary tract stones with bone mineral density in Iran, we decided to conduct this study to determine the relationship between bone density and urinary stone disease duration.
Methods
We selected 24 male patients ages between 30 to 50 years from those patients who were referred to Sina Hospital’s kidney stone removal section from March 2016 to January 2018 and were diagnosed with a recurrent urinary stone (diagnosed more than once) with imaging techniques (radiography or ultrasound). The patients had no confounding factors affecting bone mineral density (BMD), such as thyroid disease or corticosteroid usage. Patients who applied calcium restriction in their diet and those who had been in bed for a long time were excluded from the control study. To conduct an analytical case-control study, the control group, which consisted of 24 men, was selected from the patients who were referred to Sina Hospital at the same time interval and were in the same age group as the control group but without any history of urinary tract stones. The sampling method was non-random (Available samples).
Height and weight were measured for all the samples. Body mass index (BMI) was calculated by dividing the weight by the square of the height. BMD of Lumbar spine L2-L4 (BMD LS) and femoral neck (BMD FN) were measured in patients and controls using the dual-energy x-ray absorptiometry (DXA) technique and expressed in milligrams per square centimeter. The disease duration was calculated as the intervals between the first clinical presentation or the diagnosis of a stone presence until the BMD was measured and expressed in months.
Osteopenia and osteoporosis in the study group were determined according to the criterion T score, which compares BMD with the mean BMD in the control group of 20-40 years. BMD values less than (-1) and (-2/5) standard deviation were considered osteopenia and osteoporosis, respectively.
The study was under the Tehran University of Medical Sciences Ethical Committee (IR.TUMS.VCR.REC.1398.835).
We utilized SPSS-12 software, and the data were analyzed with a t-test.
Results
Table 1 shows the mean values of age, duration of urinary tract stones, and bone density in each region. The bone density in patients was 11% lower than in the control group. Femoral bone density showed a similar decrease (12%) in this group.
To evaluate the bone density in both groups, covariance analysis was used to adjust age and BMI, which are confounding variables. The difference between BMD FN and BMD LS in patients and the control group was significant (p-value<0.01). Additionally, according to covariance analysis results, BMI and age were not influenced by BMD FN and LS.
Table 1. The mean and standard deviation of age, BMI, duration of urinary stone, BMD LS, and BMD FN in patients with urinary stones and control group
|
Evaluated parameters |
Patients (Number=24) |
Control (Number=24) |
P-value |
|
Age (Years) |
38.46.7 |
39.36.8 |
-- |
|
BMI (Kg/m2) |
263.75 |
254.5 |
-- |
|
Disease duration (Months) |
262.5 |
-- |
-- |
|
BMD LS (mg/cm2) |
1018168 |
1153155 |
0.0037 |
|
BMD FN (mg/cm2) |
888117 |
1017144 |
0.0018 |
BMI: Body Mass Index; BMD: Bone Mineral Density; BMD FN: Bone Mineral Density Femoral neck; BMD LS: Bone Mineral Density Lumbar Spine.
The present study showed that 8 and 9 patients had osteopenia in the lumbar spine and neck, respectively. In the control group, these numbers were 3 and 4, respectively. There was no osteoporosis in any of the subjects. Linear regression analysis showed a significant correlation between BMD LS values and the duration of urinary stones (p-value<0.001, and r=-0.73). In the case of BMD FN, this correlation was less notable (p-value <0.01, and r=-0.52) (Figures 1 and 2).

Figure 1. Correlation of BMD Spine and duration of urinary stone in 24 patients with urolithiasis
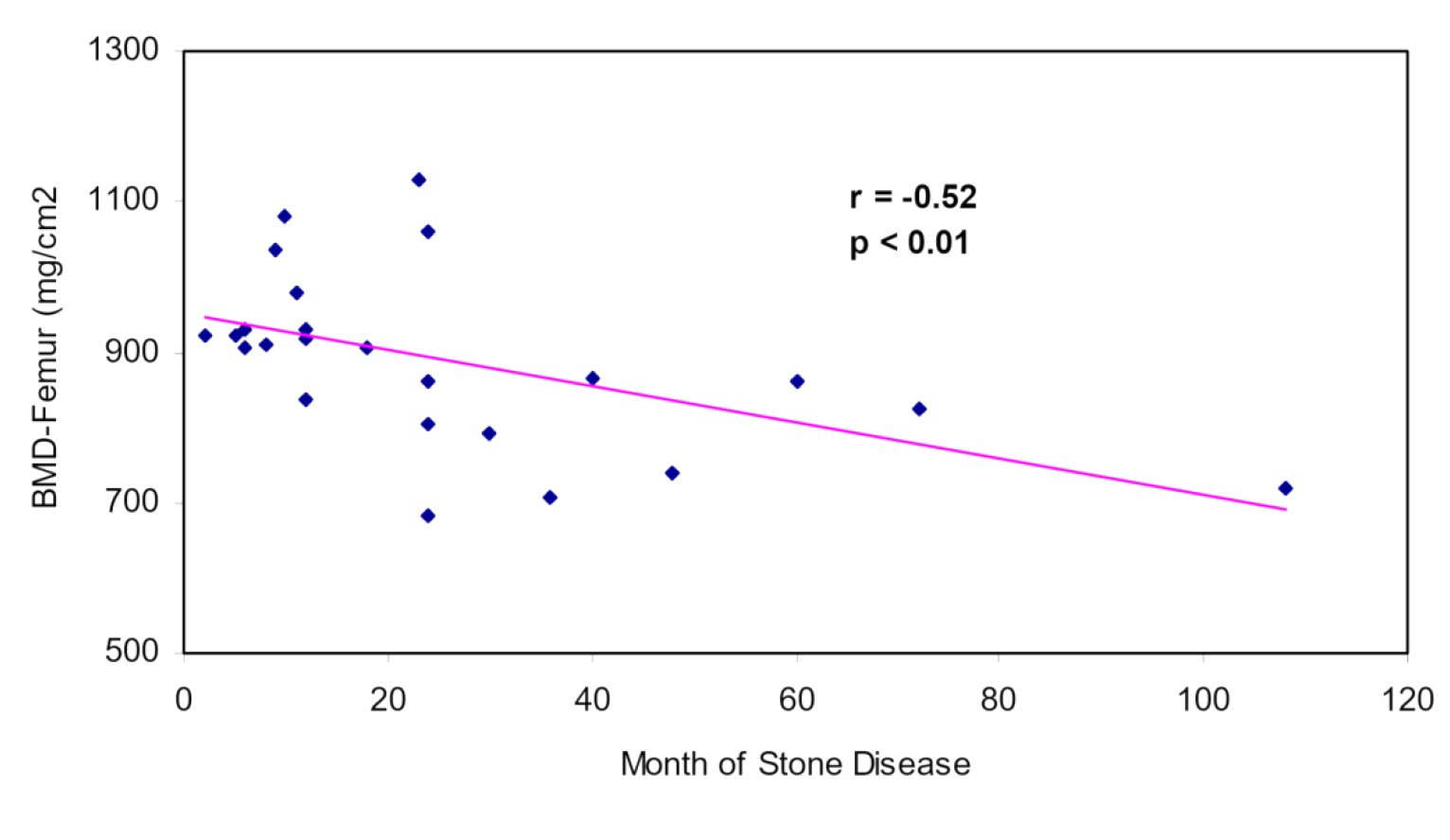
Figure 2. Correlation of BMD Femur and duration of urinary stone in 24 patients with urolithiasis
Discussion
Osteoporosis is the most common type of metabolic bone disease associated with decreased bone minerals and matrix. This disorder can be diagnosed by demonstrating typical fractures or measuring bone mineral density. Multiple techniques are available to measure bone density in the axial and appendicular skeleton, but in most patients, the DXA method is preferred for measuring BMD. The high accuracy, low radiation, low investigation time, and cost-effectiveness of DXA make this technique superior to other methods.
For the first time, Alhava et al., demonstrated a decrease in bone mineral density in patients with lithotripsy using a 2D gamma-ray attenuation technique (5). It is shown that Protease inhibitor-associated bone mineral density loss is related to hypothyroidism and related bone turnover acceleration (6).
Pietschmann F et al., measured the BMD of the lumbar spine and trunk radius of patients with urolithiasis using DXA in 1992 (7). The BMD of lumbar vertebrae was much lower in patients with hypercalciuria than in non-hypercalciuric patients. This study confirms the osteopenia of trabecular bones in patients with hypercalciuria (8).
Only male patients aged 30-50 were studied; therefore, factors affecting bone density, such as menopause or inadequate bone growth, were excluded. The results showed a significant decrease in BMD LS and BMD FN in the patients compared to the control group (p-value<0.001). This reduction appears to be mainly due to bone metabolism abnormalities in these patients.
Other studies have similarly measured bone density in patients with urinary stones and showed a significant difference in bone mineral density compared to normal subjects (9). These studies have been performed using radiological photodensitometry, micro densitometry, calcium kinetic studies, neutron activation analysis, and histomorphometric analysis (10, 11).
Non-invasive techniques that make it easier to study bone density are currently available (12). Examination of bone density was also performed by single-photon absorption technique in forearm bones (13, 14), dual photon absorption (15, 16), dual-energy radiographic absorption (17, 18), and quantitative computed tomography (19), in the lumbar spine, and showed similar results to our study. Also, some of these methods, such as dual photon absorption, are applied to classify types of idiopathic hypercalciuria (20).
Some studies showed that the history of affection to urinary stones was significantly longer in people with reduced BMD in the lumbar spine than in those with normal BMD (21, 22). We also found a similar result.
Wardle E et al., in 1985 (23) described the increase in bone turnover as the scientific justification of osteopenia in patients with urolithiasis. Moreover, the correlation between osteoporosis and calcium urolithiasis in the adult population was highlighted (24).
Based on an old belief, many people limit their consumption of dairy foods; they are at high risk for osteoporosis and its associated complications. Therefore, therapeutically limiting diary regimes in patients with urinary stones is not recommended.
Conclusions
Various references recommend metabolic examinations for all patients who are active in stone formation (which means that in a year, stone excretion has happened more than once, a second stone has been formed, or stone volume has increased). According to the result of these examinations, if calcium metabolism was impaired, bone density measurement should be performed for them. Also, their underlying disease needs to be carefully evaluated, and appropriate treatment is recommended to prevent stone formation and possible complications.
Authors’ contribution
AM was responsible for study conception and design, EHN provided data, KNJ was responsible for statistical analysis, FKH and FDT wrote the manuscript.
Acknowledgments
Special thanks to the Urology Research Center (URC), Tehran University of Medical Sciences (TUMS).
Conflict of interest
All authors declare that there is not any kind of conflict of interest.
Funding
There is no funding.
Ethical statement
The study was under the Tehran University of Medical Sciences Ethical Committee (IR.TUMS.VCR.REC.1398.835).
Data availability
Data will be provided by the corresponding author on request.
Abbreviation
BMC Bone mineral content
BMD Bone mineral density
BMI Body mass index
BMD FN Bone mineral density femoral neck
BMD LS Bone mineral density lumbar spine
DXA Dual-energy x-ray absorptiometry